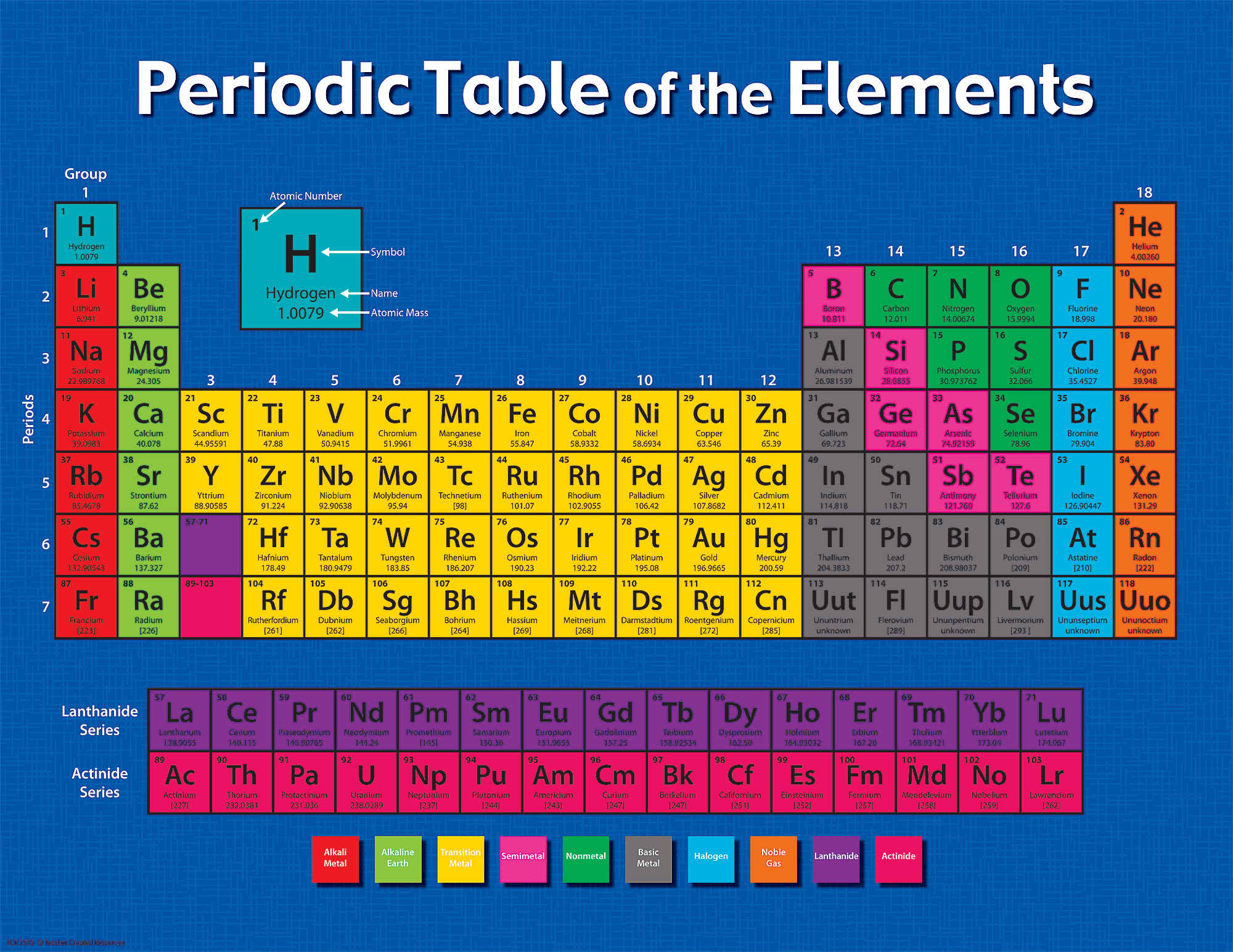 
This fact has key implications for the building up of the periodic table of elements. The ordering of the electrons in the ground state of multielectron atoms, starts with the lowest energy state (ground state) and moves progressively from there up the energy scale until each of the atom’s electrons has been assigned a unique set of quantum numbers. It is the Pauli exclusion principle that requires the electrons in an atom to occupy different energy levels instead of them all condensing in the ground state. Each electron is influenced by the electric fields produced by the positive nuclear charge and the other (Z – 1) negative electrons in the atom. The total electrical charge of the nucleus is therefore +Ze, where e (elementary charge) equals to 1,602 x 10 -19 coulombs. The number of electrons in an electrically-neutral atom is the same as the number of protons in the nucleus. The total number of protons in the nucleus of an atom is called the atomic number (or the proton number) of the atom and is given the symbol Z. In the periodic table, the elements are listed in order of increasing atomic number Z. The number of electrons in each element’s electron shells, particularly the outermost valence shell, is the primary factor in determining its chemical bonding behavior. The configuration of these electrons follows from the principles of quantum mechanics. The chemical properties of the atom are determined by the number of protons, in fact, by number and arrangement of electrons. There is a recurring pattern called the “periodic law” in their properties, in which elements in the same column (group) have similar properties. Generally, within one row (period) the elements are metals to the left, and non-metals to the right, with the elements having similar chemical behaviours placed in the same column.Įvery solid, liquid, gas, and plasma is composed of neutral or ionized atoms. It is organized in order of increasing atomic number. Element #101, discovered in 1955, was named for him, as is a crater on the far side of the moon.The periodic table is a tabular arrangement of the chemical elements. Mendeleev was nominated for a Nobel Prize, but never received one. Mendeleev studied hydrodynamics, meteorology, geology, and economics, and his interest in petroleum led to the founding of the first oil refinery in Russia. He is also credited with bringing the metric system to Russia. After meeting with international chemists at a conference in 1860, he began to worry his colleagues would fall behind with no authoritative texts in Russian, so he wrote the first Organic Chemistry textbook in that language in just 61 days. While studying to become a teacher, he began to publish original research papers, but in 1855, he was diagnosed with tuberculosis and given two years to live. He dealt with many hardships, including the death of his father when he was 13, and the loss of the family glass business when he was 15. Mendeleev’s story goes beyond his famous table. The expanded version with 118 elements is well known to all chemistry students and aficionados. His “periodic table” has become invaluable, helping to explain how elements are related, how and why they react, and why they have certain properties. In total, he predicted the existence of 10 elements, two of which were not found, due to errors in atomic weight information available at the time. He left gaps in the table where elements that fit had not yet been discovered-and he correctly predicted the properties of gallium, scandium and germanium, which had all been identified by 1889. Others published similar elemental arrangements, but his was the first. He reportedly drafted his table on Februafter a dream. This table demonstrated repeating patterns of elemental properties. 
He is most famous for finding a way to arrange chemical elements (according to their atomic weights, or the number of protons in their atomic nuclei) that resulted in a table. Dmitri Mendeleev (1834-1907) was born in Siberia, one of as many as 17 children.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
